ChemFH
In early drug discovery, detecting frequent false positives poses a significant challenge, leading to wasted time and resources in hit identification. To address this, ChemFH (Chemical Frequent Hitter), an integrated online platform, predicts potential frequent hitters across various parameters. Utilizing high-quality database and Directed Message Passing Neural Network (DMPNN) architectures, ChemFH enhances the efficiency of drug R&D by achieving reliable detection of frequent hitters in colloidal aggregate, firefly luciferase reporter enzyme inhibition, fluorescence, chemical reactivity, and promiscuity.
Calculation
Evaluation Pattern is able to comprehensively evaluate the risk of the queried molecule based on the combination of credible prediction models and useful substructure rules.
Get StartedScreening Mode is able to screen a molecular dataset and detect potential frequent hitters, thus improving the credibility of experimental results and decreasing unnecessary cost.
Get StartedMechanism Explanation

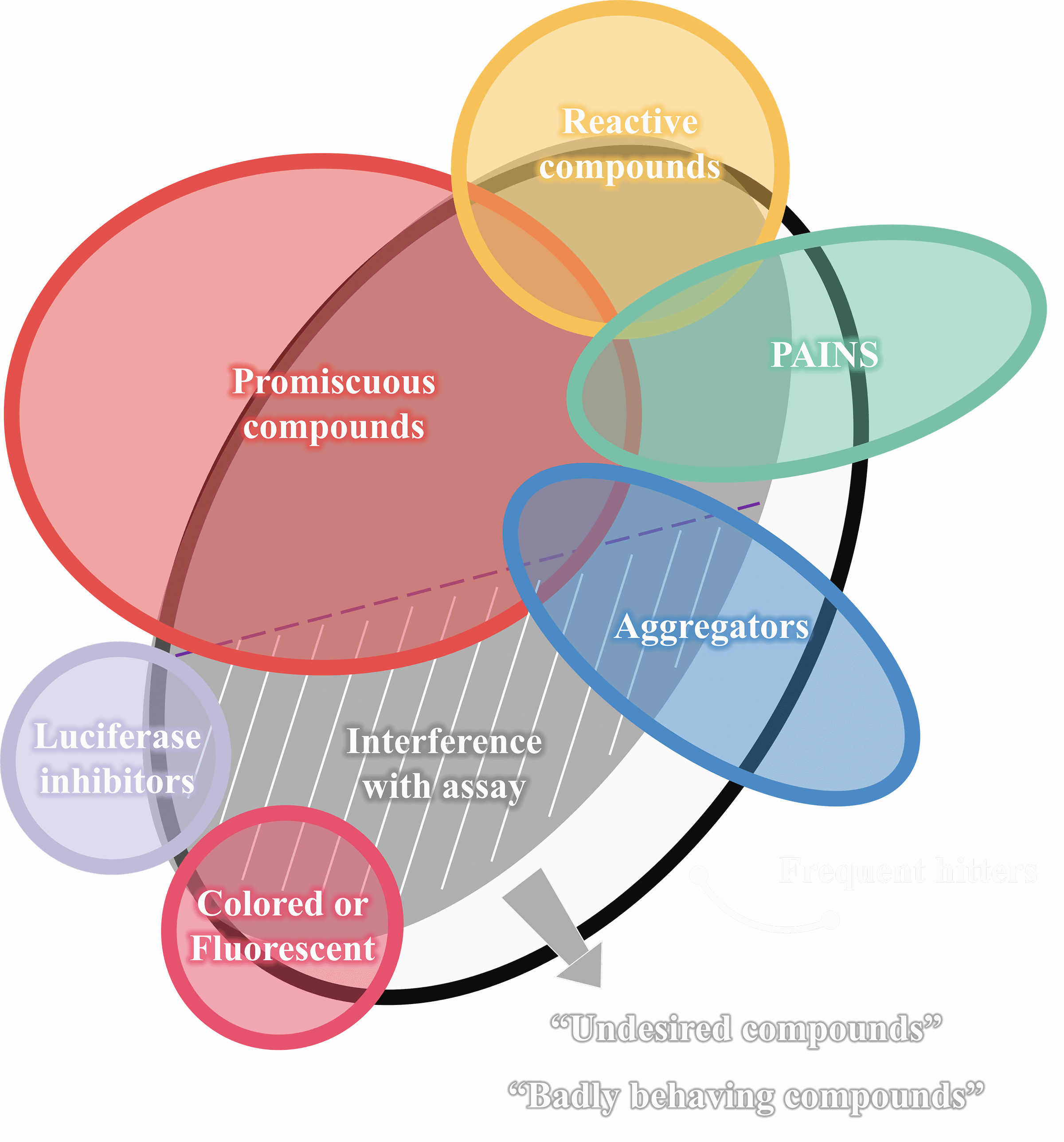
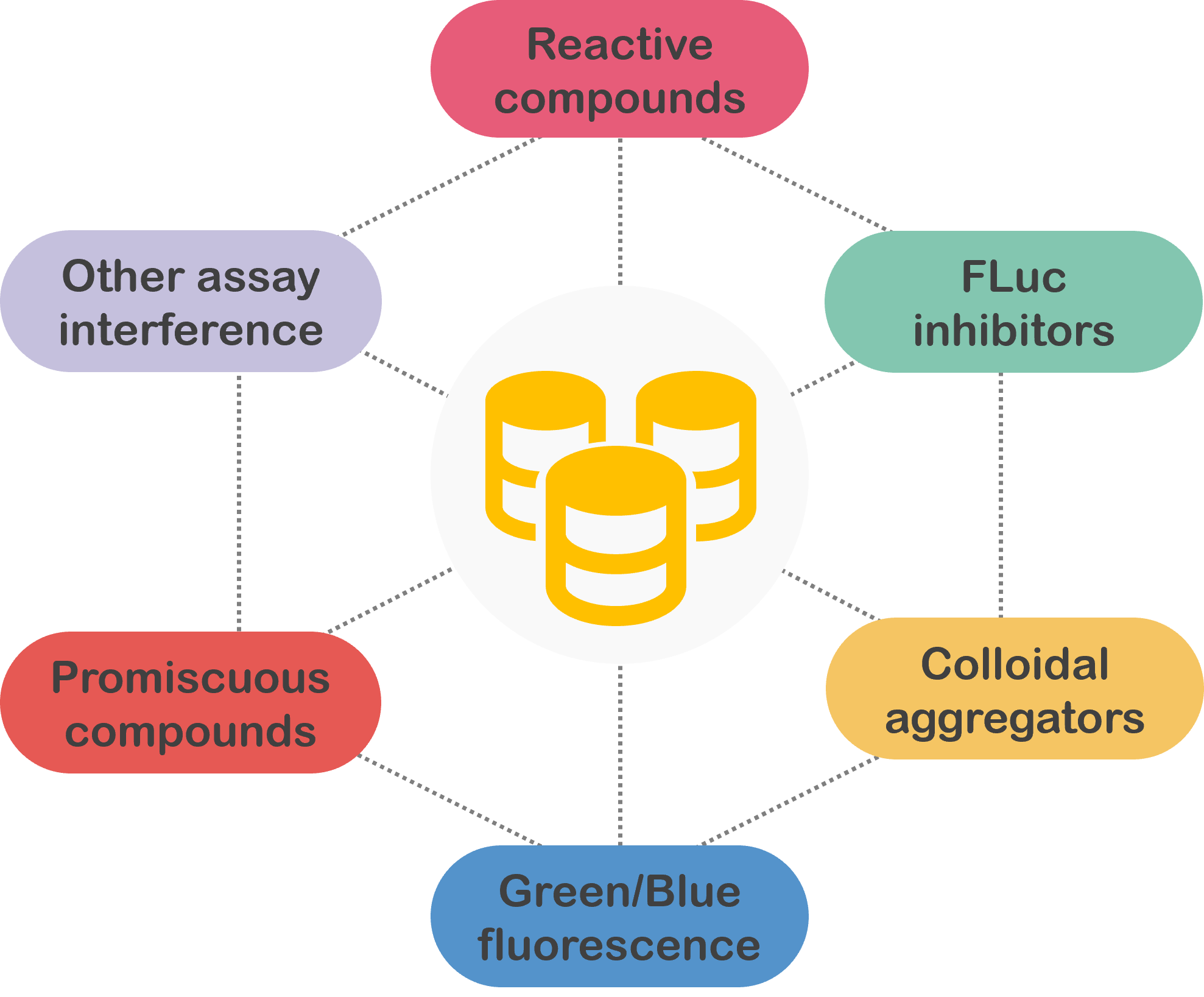
1. Colloidal aggregators
Aggregation starts when concentration exceeds the CAC, forming 30-600 nm aggregators. They non-specifically bind to protein surfaces, causing local unfolding and often resulting in enzyme destabilization or denaturation.
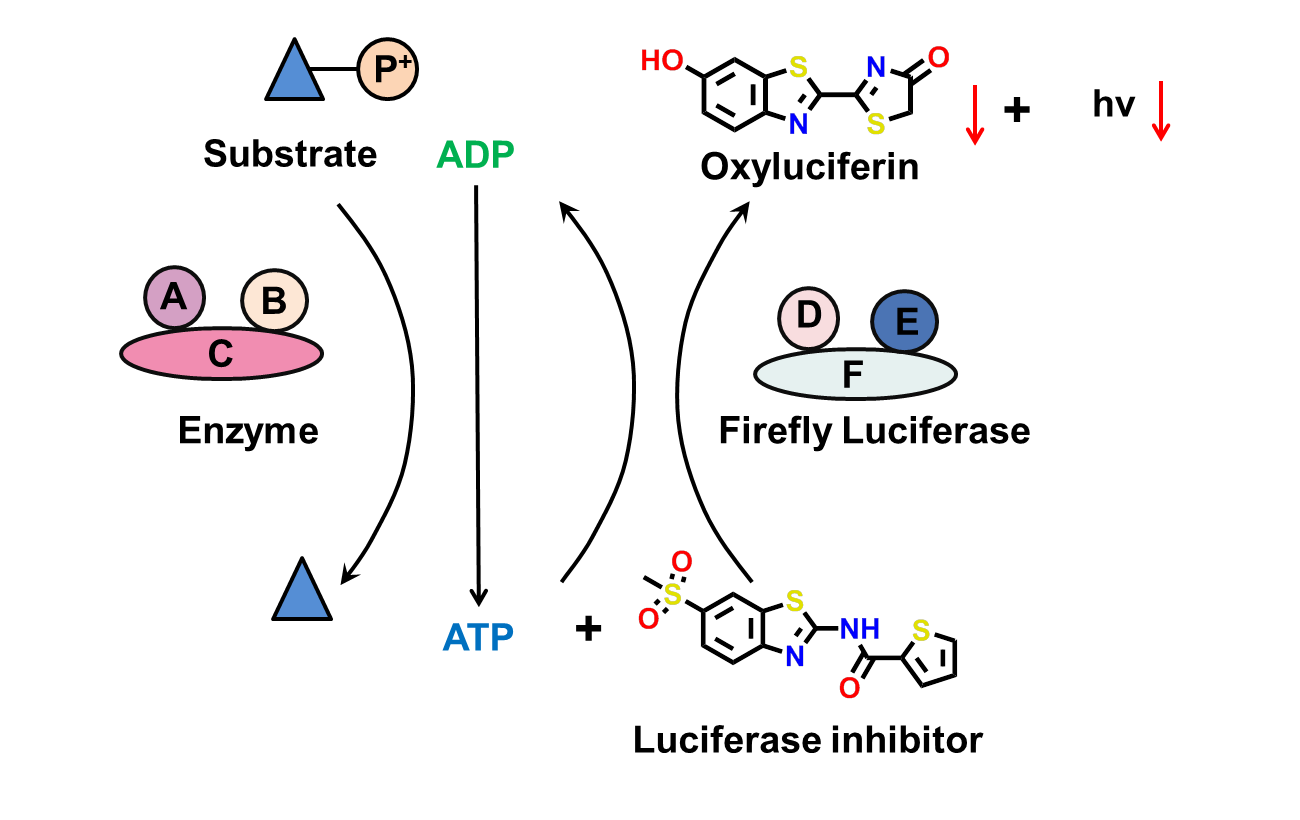
2. FLuc inhibitors
Due to its unique catalysis mechanism, firefly luciferase (Fluc) is commonly used in HTS bioluminescence assays, particularly those focused on studying gene expression at the transcriptional level. However, unexpected FLuc inhibitors can disrupt these HTS assays by inhibiting FLuc activity.
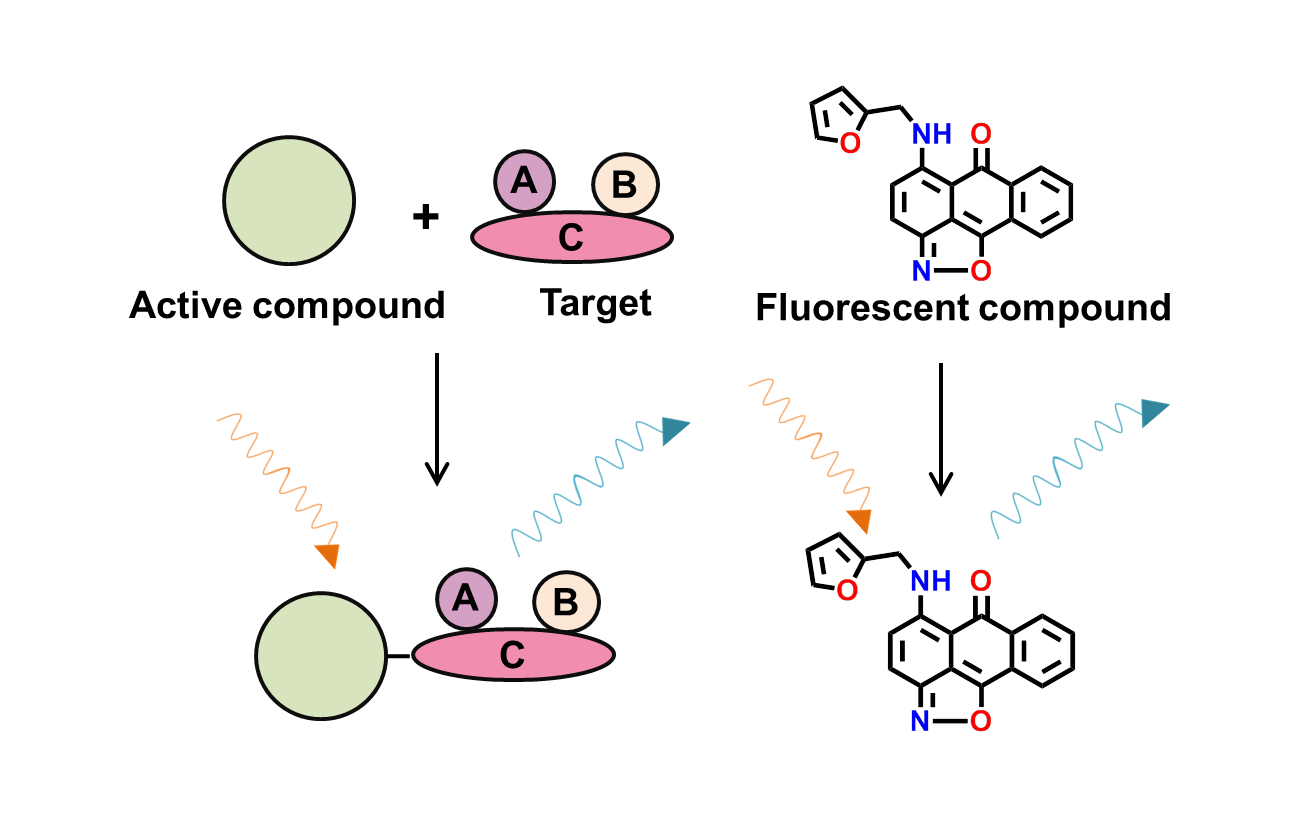
3. Blue/Green fluorescence
Fluorescence occurs when a fluorophore absorbs light, exciting an electron. Used in various applications like enzyme substrates and biomolecule labels, fluorescence can interfere with related HTS assays.
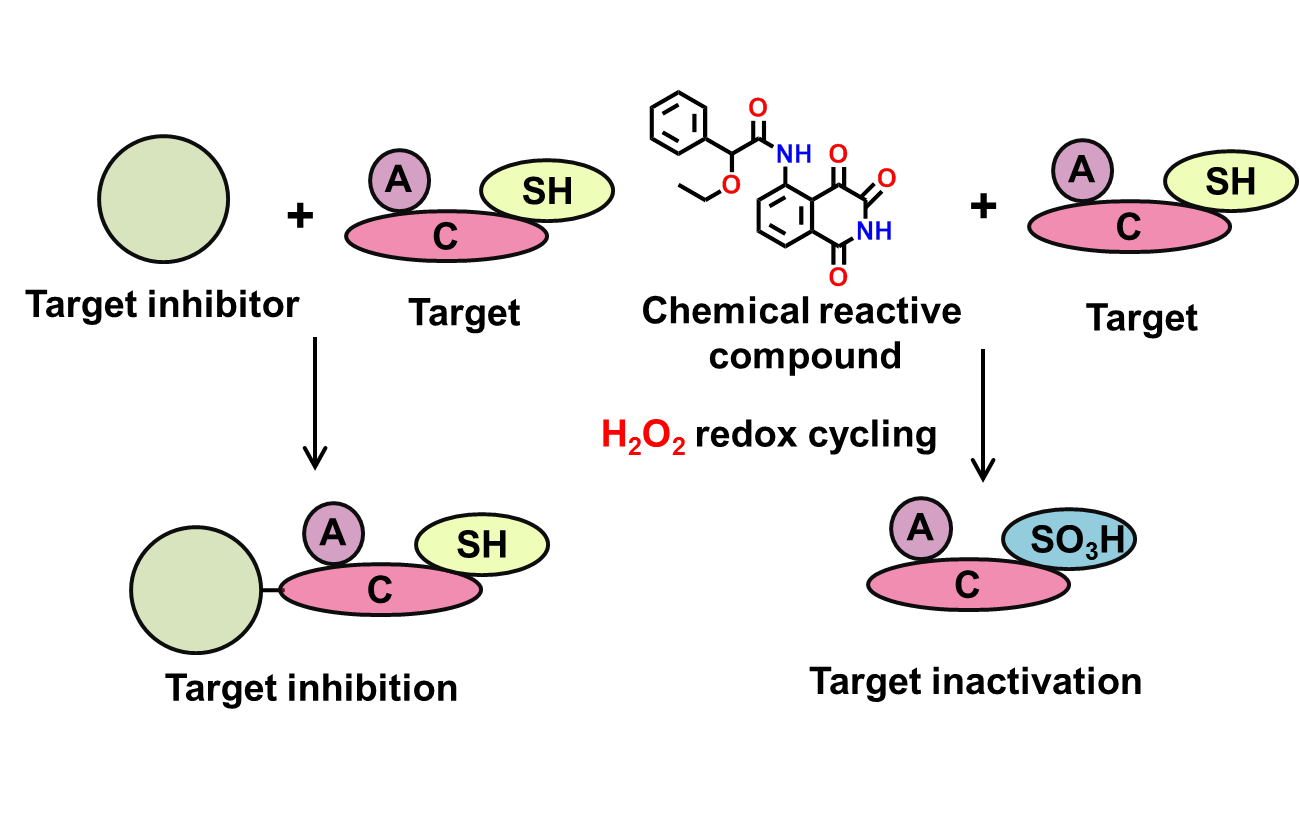
4. Reactive compounds
Chemical reactive compounds typically result in the chemical modification of reactive protein residues or, less frequently, the modification of nucleophilic assay reagents。

5. Promiscuous compounds
Promiscuous compounds refer to compounds that specifically bind to different macromolecular targets. These multiple interactions may include unintended targets, thus triggering adverse reactions and other safety issues.
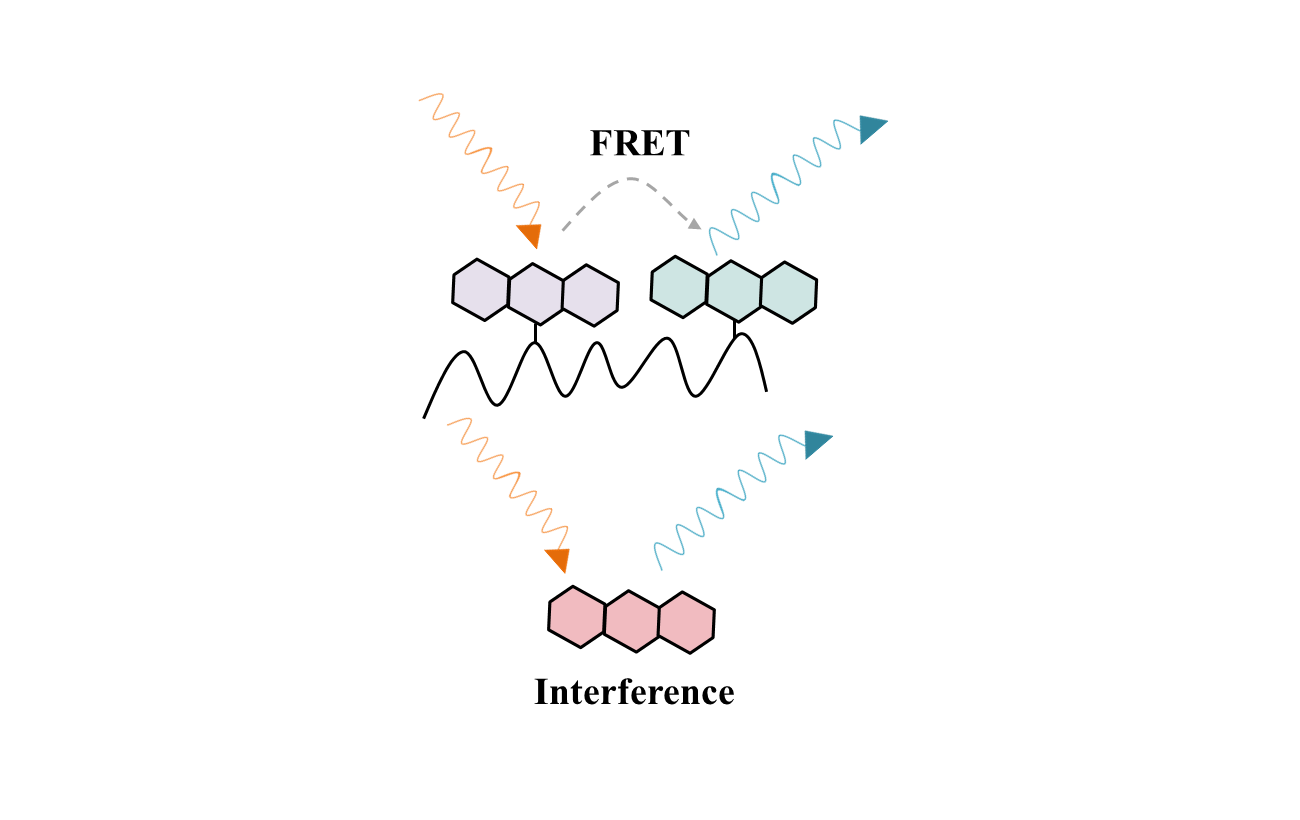
6. Other assay interferences
Alpha-screen, FRET, TR-FRET, absorbance artifacts are included.
Highlights
High-Quality Data with Clear Mechanisms
ChemFH is constructed on a dataset of over 810,000 high-quality compounds, spanning seven frequent hitter mechanisms. Sourced from literature and databases, these compounds contribute to the dataset. Clearly defining these frequent hitter mechanisms helps identify false positives misclassified as active compounds in virtual screening more accurately.
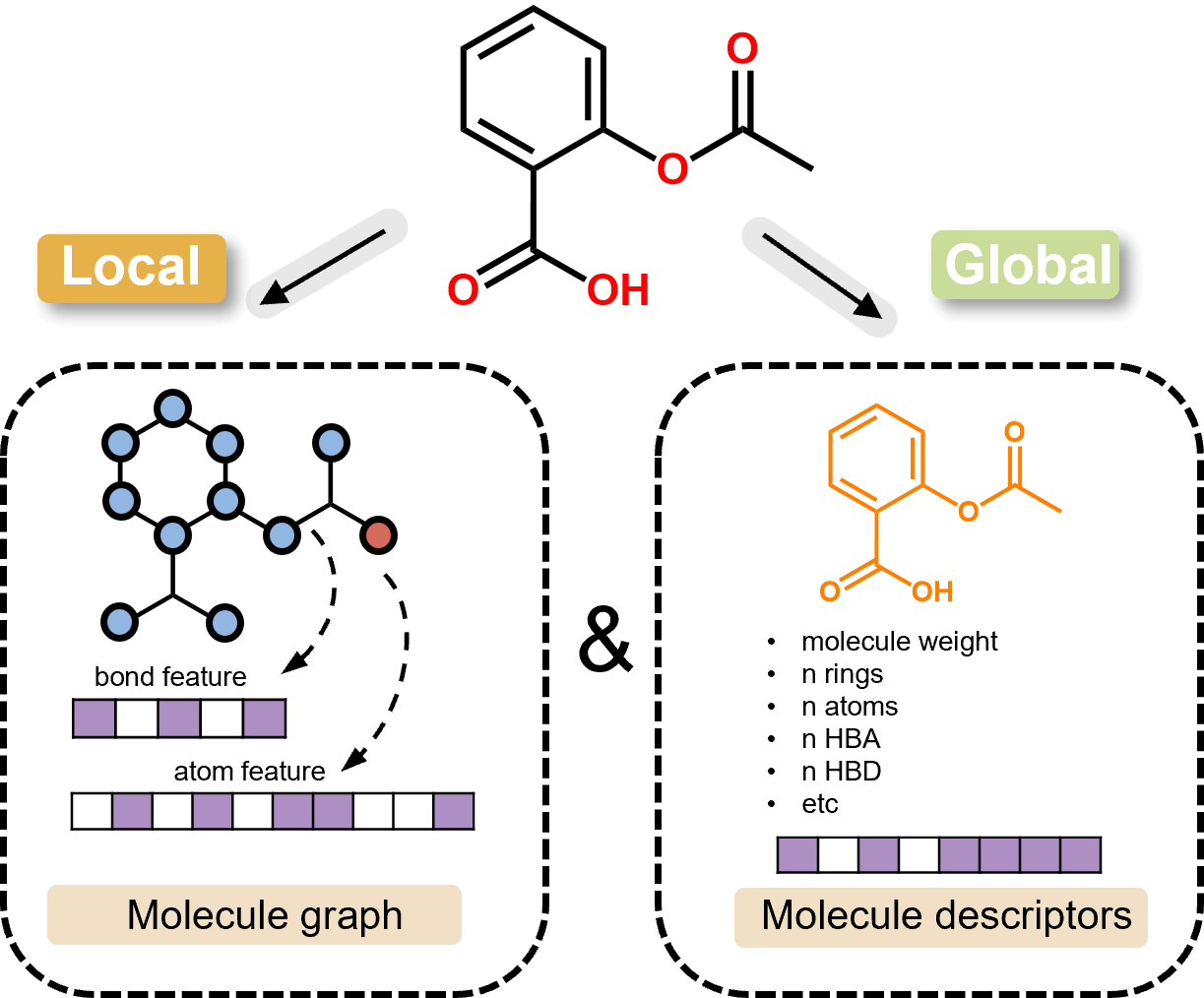
Robust and Accurate Multi-Task DMPNN Models
ChemFH utilizes the DMPNN framework, enabling message passing by fusing vectors of neighboring bonds in the molecular graph to enhance message aggregation and updating. Furthermore, the integration of molecular graph vectors with molecular descriptors significantly improves the model's performance and robustness.
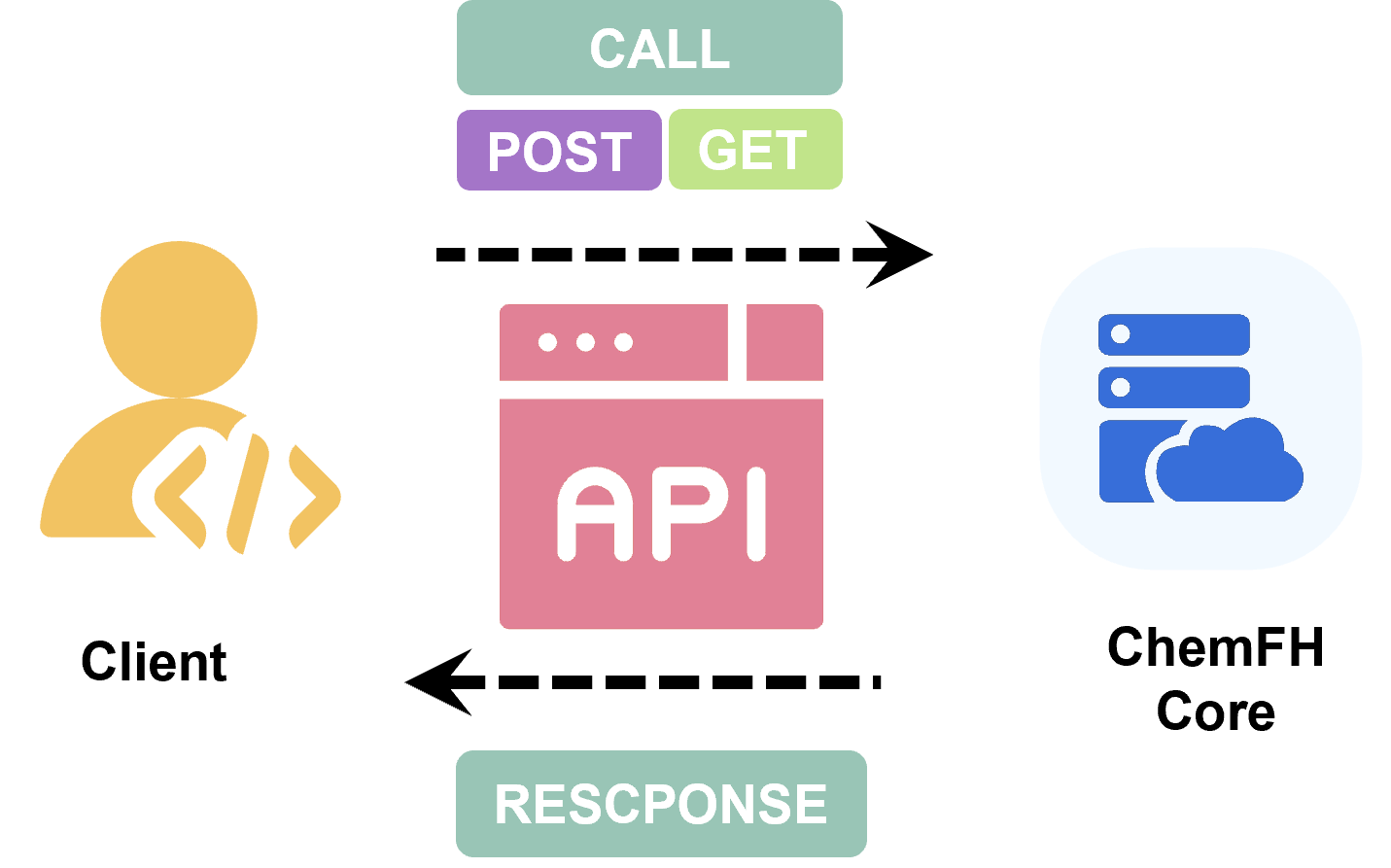
Flexible API Interfaces and User-Friendly Interaction
Flexible API interfaces designed to fulfill the need for batch calculations on extensive datasets, allowing developers to integrate its functionality into a broader range of applications or integrations. The single-molecule and multi-molecule evaluation modules are applicable to various scenarios in drug design. The interactive interface ensures user-friendly and easy usage.
Multiple FHs Alerts and Uncertainty Evaluation
The system employs ten types of frequent hitter alerts to aid in identifying structure-activity relationships of queried molecules, providing guidance for subsequent optimizations. Uncertainty estimate results are labeled as "High-confidence" or "Low-confidence" to guide the reliability of predictions for a given molecule.
This work is licensed under a Creative Commons Attribution-Noncommercial License.